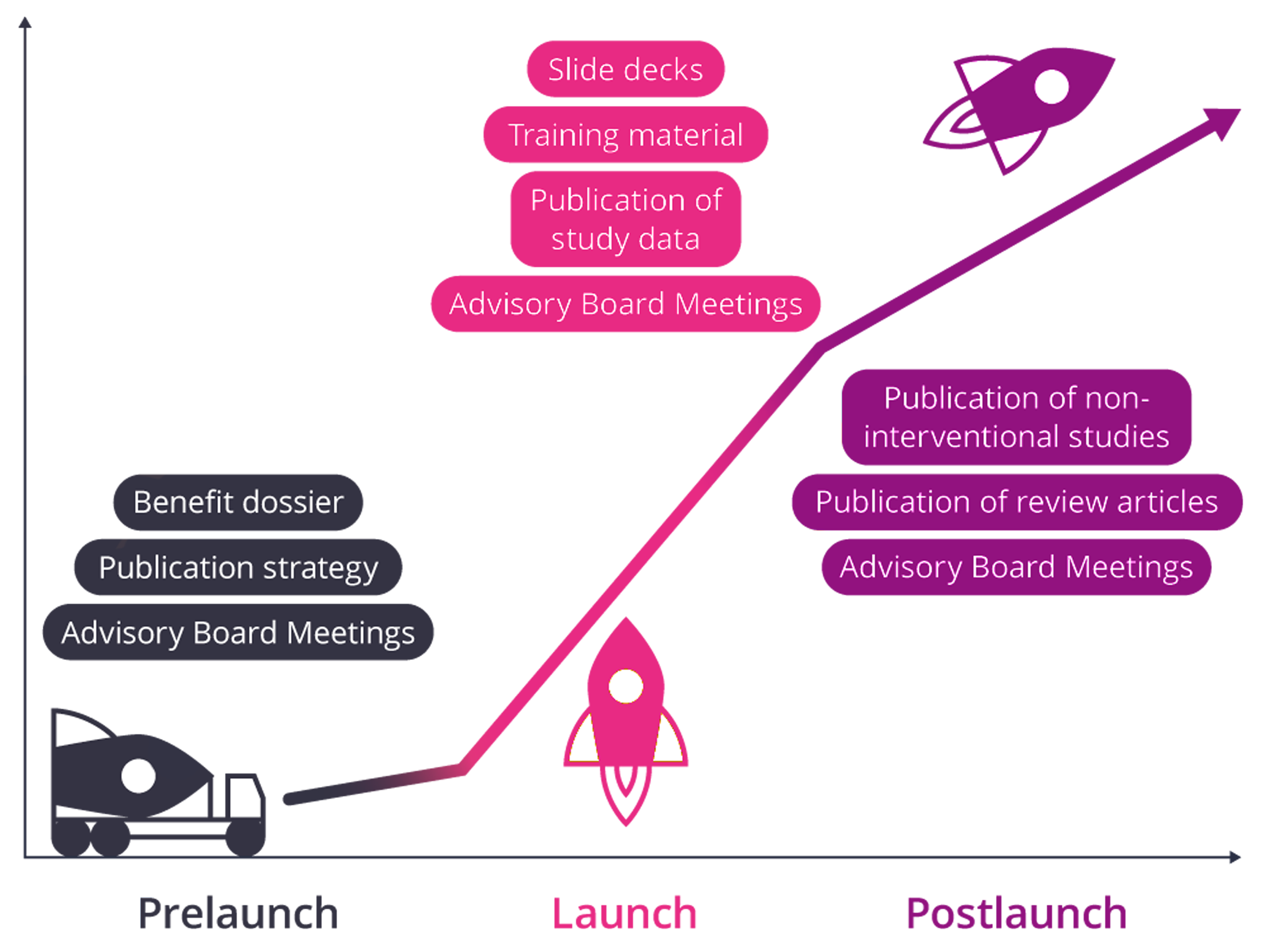
Pre-Launch
During the pre-launch phase, the most important thing is to whet the appetite for the product and optimize its positioning. Cooperation with clinical experts/KOLs is the first priority here. It is also important to have a refined publication strategy in place to ensure that study data and reviews are published in the relevant journals in a timely manner.
Our services:
- Organization, facilitation, and recording of minutes of advisory board meetings
- Development of a publication strategy
Launch
IDuring the launch phase, new data on the product are presented at congresses and questions from physicians regarding the product have to be answered. Convincing slide decks are essential here. During the launch phase, the pivotal studies are published. In addition, the sales force must be trained to become familiar with the new product.
Our services:
- Development of slide decks
- Publication of study data, preparation of study summaries, etc.
- Especially for the sales force: preparation of training documents
Post-Launch
In the post-launch phase, the product penetrates the market. New prescribers and also patients address new questions about the product to the medical affairs function, which must be answered competently and timely. The publication of reviews and of non-interventional studies anchors the product in the memory of the target group.
Our services:
- Research on medical-scientific questions
- Publication of review articles
- Publication of non-interventional studies (NIS)